
 Location: Ch 9: Gases
Location: Ch 9: Gases

 Location: Ch 9: Gases
Location: Ch 9: Gases
|
 |
GASES
IN THIS CHAPTER:
|
 ases and liquids are very similar in that they both can flow; thus they
are both called fluids. The primary difference between gases and liquids
is the distance between molecules. In a liquid, the molecules are close
together where they continually experience forces from the surrounding
molecules. In a gas, the molecules are far apart, thus allowing them to
move freely with few collisions. If two molecules collide, one may speed
up and the other slow down, but their total KE stays the same. Gases expand
to fill all available space and take the shape of its container. Gravitation
only affects gases when there is a large quantity of gas, such as around
a planet or in a star.
ases and liquids are very similar in that they both can flow; thus they
are both called fluids. The primary difference between gases and liquids
is the distance between molecules. In a liquid, the molecules are close
together where they continually experience forces from the surrounding
molecules. In a gas, the molecules are far apart, thus allowing them to
move freely with few collisions. If two molecules collide, one may speed
up and the other slow down, but their total KE stays the same. Gases expand
to fill all available space and take the shape of its container. Gravitation
only affects gases when there is a large quantity of gas, such as around
a planet or in a star.
| Gas | Temperature | Density (kg/m3) |
| Dry air |
0°C
|
1.29 |
|
10°C
|
1.25 | |
|
20°C
|
1.21 | |
|
30°C
|
1.16 | |
| He | 0.18 | |
| H | 0.09 | |
| O2 | 1.43 |
On Earth, gases can be found almost everywhere. Earth’s atmosphere is a large ocean of different gases. These molecules are kept in continual motion by the sun’s rays and are prevented from flying into outer space by gravitational force. Unlike an ocean, however, the atmosphere does not have a definite surface, and its density decreases in higher altitudes. Thus the atmosphere is very much like a huge pile of feathers. Eventually the air thins out until there is no air at all (and then you’re in space.) Although the atmosphere goes nearly 40km above the earth’s surface, 50% of the atmosphere is below 5.6km in atmosphere. 75% of the atmosphere is below 11km and 99% is below an altitude of 30km. The temperature at the highest edge of the atmosphere is actually hotter than some of the air below it. At first, that may seem strange, but ultraviolet light strikes the ozone layer at the top of the atmosphere. The light splits apart the O3 atoms into O2 atoms and heats
We live at the bottom of an ocean of air. Since atmospheric pressure is caused by the weight of the air above it, we live in relatively high air pressure. We are so accustomed to it, however, that we tend to forget that it has weight. At sea level, the force being exerted on 1 square meter is about 101.3kPa.
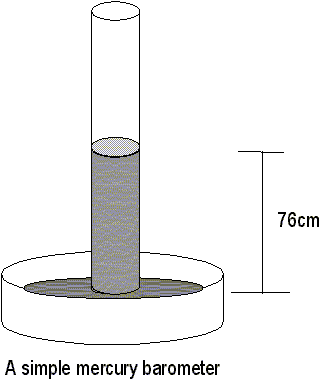
Barometers are instruments that are used to measure the pressure of the atmosphere. A 100cm glass tube is filled to the brim with mercury and closed at one end. It is then tipped upside-down into an open dish of mercury. The mercury inside the tube will run into the dish until the level of mercury inside the tube is about 76cm. It may seem strange that the mercury runs out the bottom of the tube, but the explanation is simple. The mercury inside the barometer stays at the top when the weight of the liquid in the tube is equal to (or less than) the weight of the air outside. When the weight of the liquid is too great, the mercury falls into the dish, leaving a near-vacuum of mercury vapor behind. Since mercury has a density 13.6 times that of water, a barometer of water would have to be 10.3m (13.6´0.76m) in height. This also explains why the bottom of a simple water pump cannot be deeper than 10.3m.
Another type of barometer that does not use mercury is the aneroid barometer. Such a barometer employs a small metal box that is partially exhausted of air and has a flexible lid that bends in and out with the outside air pressure. A lever is attached to the lid of the small box and held in place by a spring. This lever rotates a needle on the outside of the barometer and indicates the current air pressure. Since air pressure decreases with elevation, a barometer can be used as an altimeter.
Drinking through a straw is similar to the operation of a barometer. When you drink through a straw, you are not sucking on the straw. To get the drink into your mouth requires that you create a partial vacuum inside the straw. Your diaphragm pulls down on your lungs, thus increasing your lungs’ volume. This increase of volume de-pressurizes the air inside your lungs and the straw. Since the air inside the straw no longer pushes the fluid down, the atmosphere outside pushes the drink up through the straw and into your stomach.
 The air pressure inside a bicycle tire is considerably greater than the
air pressure outside. The density is also considerable greater. Suppose
the number of air molecules inside this tire is doubled. If the air has
the same temperature, then the air density will be doubled and there will
be twice as many collisions between the air molecules. This means that
the pressure inside is also doubled. Therefore, pressure is directly proportional
to density. The density of air can also be doubled by simply compressing
the air to half its volume. This means that pressure ´
volume = 1. As long as the temperature does not change, and no molecules
escape out of the container, the following equation is also true: P1V1
= P2V2
The air pressure inside a bicycle tire is considerably greater than the
air pressure outside. The density is also considerable greater. Suppose
the number of air molecules inside this tire is doubled. If the air has
the same temperature, then the air density will be doubled and there will
be twice as many collisions between the air molecules. This means that
the pressure inside is also doubled. Therefore, pressure is directly proportional
to density. The density of air can also be doubled by simply compressing
the air to half its volume. This means that pressure ´
volume = 1. As long as the temperature does not change, and no molecules
escape out of the container, the following equation is also true: P1V1
= P2V2
There are two types of air pressure gauges. Absolute pressure gauges measure from 0kPa (that is, a perfect vacuum) and must be calibrated in a vacuum. Other air pressure gauges, such as those for measuring the pressure in car tires read 0lb/in2 but actually are reading 14.7lb/in2. In order to get a true measurement of air pressure inside the tire, you need to add 14.7lb/in2.
Buoyancy was discussed in the previous section entitled "Liquids". All the rules for buoyancy were stated in terms of fluids rather than liquids because the rules hold for gases as well. Thus a dirigible "floats" in the air for the same reason as a fish does.
So far, this paper has only discussed stationery fluids. Motion produces an additional influence. Most people think that atmospheric pressure increases during hurricanes, and thus people’s roofs blow off. Actually, the opposite is true. The air inside the attic of the house is actually greater than that of the air outside. This strange occurrence is called Bernoulli’s principle, named after the 18th century scientist Daniel Bernoulli. In a pipe, the amount of water (or air) that flows past any given point is always the same. Since the amount of fluid that flows past a given point inside a point is always the same, regardless of the pipe’s width, the pressure exerted on the walls of the pipe must change. Thus, if a pipe constricts, the water pressure will increase greatly.
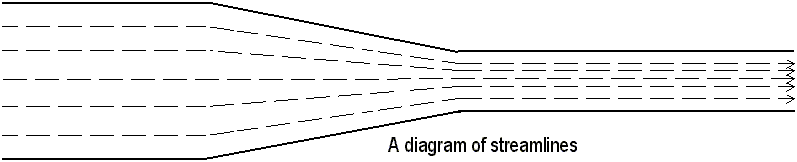

Bernoulli’s Principle is also involved in curve balls. When a moving ball spins, unequal air pressures are produced on opposite sides of the ball. These unequal pressures result in additional weight or lift on the ball, and it curves up or down. The same thing happens when you turn on a hairdryer and place a foam ball over the hairdryer’s opening. The air from the hairdryer has a lower pressure than the atmosphere, and thus it pushes the ball into the path of the air from the hairdryer.
In conclusion, a gas is much like a liquid except that it expands to fit its container. The atmosphere of the earth extends about 40km above the surface of the earth. Air is more compressed at sea level than at higher altitudes. The air pressure at sea level is 101.3kPa. Simple barometers measure atmospheric pressure by measuring how high a column of mercury can be supported by atmospheric pressure. Aneroid barometers work by measuring the position of a moveable lid against a box containing low-pressure air. Boyle’s law states that at constant temperature, the pressure of a fluid is inversely proportional as its volume; if one increases, the other decreases. Bernoulli’s principle states that the pressure of a fluid flowing horizontally decreases as the speed of the fluid increases. Bernoulli’s principle explains lift and works only for steady flow.